Flow Meter Volumetric and Mass Flow Rate – What’s the Difference and How to Choose
In modern industries, accurate flow measurement is critical for controlling production, maintaining quality, and improving efficiency. Whether in oil and gas, chemical processing, food production, or water treatment, understanding how much fluid is moving through a pipeline is essential.
Two key parameters describe flow: volumetric flow rate and mass flow rate. Although they sound similar, they represent different concepts and are measured using different types of flow meters. Choosing between volumetric flow meters and mass flow meters depends on your process conditions, flow meter principle, the type of fluid, and what data you need.
This article explains the difference between volumetric and mass flow rate, the principles behind each measurement, the types of flow meters available, and how to select the right one for your application.
What Is Volumetric Flow Rate?
The volumetric flow rate measures the volume of fluid passing through a pipeline per unit of time. It tells you how much space the fluid occupies, not how much it weighs.
The common units are:
• Cubic meters per hour (m³/h)
• Liters per minute (L/min)
• Gallons per minute (GPM)
For example, if water flows at 10 GPM, it means 10 gallons of water pass through the pipe every minute — regardless of temperature or pressure.
Formula:
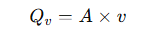
Where:
• Qv = volumetric flow rate
• A = pipe cross-sectional area
• v = average flow velocity

Volumetic flow unit L/m ( liter per minute)
Volumetric flow measurement is straightforward, but it doesn’t consider changes in density, which can vary with temperature, pressure, or fluid composition.
For liquids with stable density (like water at room temperature), volumetric flow meters are sufficient. But for compressible fluids (like gases or steam), or liquids whose density changes with temperature, volumetric measurement can be misleading.
What Is Mass Flow Rate?

Mass flow unit kg/min
The mass flow rate measures the actual mass (or weight) of the fluid passing through a pipe per unit of time. It tells you how much material is being transferred — which is crucial when dealing with chemical reactions, combustion, or blending processes.
The common units are:
• Kilograms per hour (kg/h)
• Tons per hour (t/h)
• Pounds per minute (lb/min)
Formula:
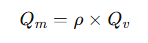
Where:
• Qm= mass flow rate
• ρ= fluid density
• Qv = volumetric flow rate
This equation shows that mass flow depends on density. If temperature or pressure changes alter density, the mass flow will remain constant while volumetric flow changes.
For example, 1 m³ of hot water weighs less than 1 m³ of cold water. But if you measure by mass (say 1000 kg/h), the total material is always the same, regardless of temperature.
That’s why mass flow meters are preferred for applications where precise material balance or chemical dosing is required.
Why the Difference Matters
In many industries, processes depend on mass, not volume. A reactor needs the correct mass ratio of ingredients. A boiler must receive the right mass of fuel and air. And gas custody transfer (such as LPG, LNG ) is based on mass or standardized volume (at reference temperature and pressure).
If you use a volumetric flow meter in conditions where temperature or pressure fluctuates, your readings can be inaccurate. The same pipeline could show different flow rates simply due to density changes.
In contrast, a mass flow meter automatically accounts for density variation, providing consistent, accurate results even under changing conditions.

For gas custody transfer ,mass flow meter is needed.
Volumetric Flow Meters: Working Principles and Types
Volumetric flow meters measure how much space the fluid occupies in the pipeline. They are ideal for clean, stable liquids or when only volume is needed. Below are common types:
1. Electromagnetic Flow Meter (Mag Meter)
Works on Faraday’s Law of Electromagnetic Induction. As conductive fluid passes through a magnetic field, it generates a voltage proportional to velocity.
Best for: Conductive liquids such as water, wastewater, chemicals.
Advantages: No moving parts, high accuracy, wide range.
Limitations: Cannot measure non-conductive fluids like oil or gas.
2. Turbine Flow Meter
Uses a rotating turbine; the rotation speed is proportional to fluid velocity.
Best for: Clean, low-viscosity liquids (water, fuels, light oils).
Advantages: High accuracy, good repeatability.
Limitations: Not suitable for dirty or viscous fluids.
3. Positive Displacement Flow Meter
Measures flow by trapping fixed volumes of fluid and counting cycles.
Best for: High-viscosity liquids (lubricants, syrups, greases).
Advantages: Measures low flow rates with excellent precision.
Limitations: Moving parts require maintenance.
4. Ultrasonic Flow Meter
Measures flow using sound waves. Transit-time type works for clean liquids; Doppler type for dirty or aerated fluids.
Best for: Large pipes, non-intrusive installations.
Advantages: No pressure loss, easy installation.
Limitations: Accuracy depends on pipe and fluid condition.
Mass Flow Meters: Working Principles and Types
Mass flow meters directly measure the mass of fluid passing through, independent of density changes. Here are the most common types:
1. Coriolis Mass Flow Meter
Based on the Coriolis effect, it measures the twisting of vibrating tubes caused by fluid mass flow.
Best for: Liquids, slurries, and gases.
Advantages: Measures mass, density, and temperature simultaneously; very high accuracy (±0.1%).
Limitations: Higher cost, larger size for big pipes.
2. Thermal Mass Flow Meter
Measures the amount of heat absorbed by the fluid as it passes a heated sensor. The heat transfer is proportional to mass flow.
Best for: gases such as air, nitrogen, natural gas.
Advantages: Direct mass flow measurement, low pressure drop.
Limitations: Not suitable for liquids.
3. Correlated (Inferential) Mass Measurement
Sometimes, volumetric flow is measured and converted to mass using separate temperature and pressure sensors.such as vortex flow meters, gas turbine flow meters .
While cheaper, this indirect method introduces additional uncertainty.
Related products
Of course, sometimes we can also set a fixed density value in the flow meter to display the mass flow rate directly, such as in kg/hr or lb/min. However, this method is not very accurate, because the density remains constant and does not change with actual process conditions.
Comparing Volumetric vs. Mass Flow Meters
|
Parameter
|
Volumetric Flow Meter
|
Mass Flow Meter
|
|
Measured Value
|
Volume (m³/h, GPM)
|
Mass (kg/h, lb/min)
|
|
Dependence on Density
|
Affected by density
|
Independent of density
|
|
Temperature/Pressure Sensitivity
|
High
|
Low
|
|
Typical Fluids
|
Liquids
|
Liquids & Gases
|
|
Accuracy
|
±0.5–1%
|
±0.1–0.2% or higher for gas (±0.5–1% )
|
|
Cost
|
Lower
|
Higher
|
|
Maintenance
|
Simple
|
Moderate
|
|
Best Applications
|
Water, oils, stable liquids
|
Gas, steam, chemical dosing
|
Choosing Between Volumetric and Mass Flow Measurement
When selecting a flow meter, consider the following factors:
1. Fluid Type – Is it a liquid,
gas, or multiphase fluid?
2. Process Conditions – Are pressure or temperature stable?
3. Measurement Purpose – Do you need total mass, or just volume?
4. Accuracy Requirements – How critical is measurement precision?
5. Budget and Maintenance – Balance accuracy vs. cost.
When to Use a Volumetric Flow Meter:
• You are measuring incompressible liquids with stable density (e.g., water).
• You need cost-effective flow monitoring.
• You want local flow indication without mass conversion.
When to Use a Mass Flow Meter:
• You are dealing with compressible fluids (gases, steam).
• Temperature or pressure varies significantly.
• You need mass balance, chemical dosing, or energy measurement.
Real-World Examples
• Water Treatment Plants: Use electromagnetic flow meters for volumetric measurement since water density is stable.
• Oil & Gas: Use Coriolis mass flow meters for custody transfer, as mass is constant regardless of temperature.
• Compressed Air Systems: Use thermal mass flow meters to monitor consumption and leaks.
• Food Industry: Use positive displacement meters or liquid mass flow meter for viscous fluids like syrup,honey,milk,juice, measured by volume.
Output and Integration
Both volumetric and mass flow meters can provide
digital signals such as:
• 4–20 mA current output
• Pulse output for totalizing
• MODBUS RTU / HART / RS485 communication /PROFIBUS DP
These allow easy integration into PLC or SCADA systems, enabling remote monitoring, data logging, and process control.
Conclusion
Understanding the difference between volumetric and mass flow rate is crucial for accurate flow measurement and process optimization.
• Volumetric flow rate tells you how much space the fluid occupies, ideal for stable liquids.
• Mass flow rate tells you how much matter is moving, ideal for gases or changing conditions.
Each flow meter type — whether electromagnetic, turbine, Coriolis, or thermal mass — serves specific needs. The right choice depends on your fluid type, process conditions, and measurement goals.
By selecting the appropriate flow meter technology, industries can ensure accuracy, reduce waste, and improve efficiency — from water treatment to oil refining, from chemical dosing to gas distribution.
Choosing wisely means measuring smarter, saving costs, and keeping operations under full control.
 English
English






 Coriolis Mass fuel flow meter
2020/07/03
Coriolis flow meter is one of the most accurate flow instruments and it has been used by different industries,it is also a perfect and excellent instruments to be used as mass fuel flow meter.
VIEW
Coriolis Mass fuel flow meter
2020/07/03
Coriolis flow meter is one of the most accurate flow instruments and it has been used by different industries,it is also a perfect and excellent instruments to be used as mass fuel flow meter.
VIEW
 Electronic mass flow meter
2020/07/03
Electronic mass flow meter is used todetermine the rate of flow of liquids and gasses. This is essential indifferent areas of plant production, efficiency and quality. For instance,since the law of co...
VIEW
Electronic mass flow meter
2020/07/03
Electronic mass flow meter is used todetermine the rate of flow of liquids and gasses. This is essential indifferent areas of plant production, efficiency and quality. For instance,since the law of co...
VIEW
 Air flow rotameter
2020/07/03
This air flow rotameter also termed as variable area air flow meter is a commercial instrument that functions asa flow measuring device. It is an accurate and precise device that specifies inhazardou....
VIEW
Air flow rotameter
2020/07/03
This air flow rotameter also termed as variable area air flow meter is a commercial instrument that functions asa flow measuring device. It is an accurate and precise device that specifies inhazardou....
VIEW
 Electronic rotameter
2020/07/03
Electronic rotameter meets modern industrydemand to have digital flow indicator also with various outputs for different processcontrol. These rugged electronic metal meters are mechanical devices that...
VIEW
Electronic rotameter
2020/07/03
Electronic rotameter meets modern industrydemand to have digital flow indicator also with various outputs for different processcontrol. These rugged electronic metal meters are mechanical devices that...
VIEW
 Volumetric Flow Meter
2020/07/03
In the past, the farming system waspracticed in areas that received considerable rainfall in order to enhanceplant growth. With volumetric flow meters invention, farming has become easy asthey ensure...
VIEW
Volumetric Flow Meter
2020/07/03
In the past, the farming system waspracticed in areas that received considerable rainfall in order to enhanceplant growth. With volumetric flow meters invention, farming has become easy asthey ensure...
VIEW
 Turbine flow meter stainless steel
2020/07/03
Turbine flow meter is an electronic flow measurementinstrument that provides the function of measurement of fluid flow rate. Theprecision and accuracy of this voltage and pulse flow sensing device mak...
VIEW
Turbine flow meter stainless steel
2020/07/03
Turbine flow meter is an electronic flow measurementinstrument that provides the function of measurement of fluid flow rate. Theprecision and accuracy of this voltage and pulse flow sensing device mak...
VIEW
 Turbine air flow meter
2020/07/03
Air turbine flow meter overviewTurbine air flowmeter is almost independentof air density, pressure, temperature and other parameters when measuring thevolumetric air flow.
VIEW
Turbine air flow meter
2020/07/03
Air turbine flow meter overviewTurbine air flowmeter is almost independentof air density, pressure, temperature and other parameters when measuring thevolumetric air flow.
VIEW
 Liquid Mass flow meter
2020/07/03
All kinds liquid measurement Density measurement as well Fuel, syrup, polymer, molasses Also, can measure corrosive liquid
VIEW
Liquid Mass flow meter
2020/07/03
All kinds liquid measurement Density measurement as well Fuel, syrup, polymer, molasses Also, can measure corrosive liquid
VIEW
 Natural Gas Flow Meter
2020/07/03
Thermal mass flow meter . Low pressure gas measurement. No moving parts flow sensor. 4-20mA, MODBUS -RTU.
VIEW
Natural Gas Flow Meter
2020/07/03
Thermal mass flow meter . Low pressure gas measurement. No moving parts flow sensor. 4-20mA, MODBUS -RTU.
VIEW
 Mass flow meters for gas
2020/07/03
At present, the main gas flow meter is a volume gas flow meter, and when the temperature and pressure change, the volumetric flow meter measurement accuracy will become poor. The advantage of the gas...
VIEW
Mass flow meters for gas
2020/07/03
At present, the main gas flow meter is a volume gas flow meter, and when the temperature and pressure change, the volumetric flow meter measurement accuracy will become poor. The advantage of the gas...
VIEW
 Large size Coriolis mass flow meter
2020/07/03
Large size: 4 inch,5 inch, 6 inch, 8 inch, 10 inch,12 inch. Max flow range: 1000 ton/hour. Option with high pressure design. Direct mass flow crude oil, fuel and so on.
VIEW
Large size Coriolis mass flow meter
2020/07/03
Large size: 4 inch,5 inch, 6 inch, 8 inch, 10 inch,12 inch. Max flow range: 1000 ton/hour. Option with high pressure design. Direct mass flow crude oil, fuel and so on.
VIEW
 Vortex Flow Meter
2020/07/03
Vortex Flow meter is for clean liquid, gas and steam flow measurement. Contact us to choose proper type flow sensor and get the competitive quotation.
VIEW
Vortex Flow Meter
2020/07/03
Vortex Flow meter is for clean liquid, gas and steam flow measurement. Contact us to choose proper type flow sensor and get the competitive quotation.
VIEW